|
These trends exist because of the similar electron configurations of the elements within their respective groups or periods they reflect the periodic nature of the elements. For hydrogen, there is only one electron and so the nuclear charge ( Z) and the effective nuclear charge ( Z eff) are equal. Major periodic trends include atomic radius, ionization energy, electron affinity, electronegativity, valency and metallic character. This is the pull exerted on a specific electron by the nucleus, taking into account any electron–electron repulsions. This activity can stand alone or be used with the attached worksheet and/or create other worksheets when discussing atom radius or ion radius.\). It helps them learn it better than reading it off another chart and it gives them ownership so they keep them longer. Have students add information to the back of their tables like polyatomic ions, equations, key concepts, exceptions to rules. Have students reference their tables when introducing concepts like atomic and ionic radius trends, ionic and covalent bonding regions. This works best when either all the information is on the table or when discussing properties. Information that could be included, but is not limited to, are element name, atomic numbers, mass numbers or weights, common oxidation numbers.Īt some point the columns could be lightly shaded and a legend could be added. The major Periodic trends in periodic table are Valency trend. This activity has students place the electron-dot model in each appropriate box, but if boxes are large enough other information maybe added throughout the year. Make sure students are filling in the boxes correctly from the start! Have students keep their tables for a reference tool. The valence electrons are symbolized as a dot and placed in increasing number around the symbol.Įven though the numbering scheme is no longer widely used it will be helpful later when introducing the idea of half-filled S and P sub-orbital's. Where X is the symbol of the element and accounts for the nucleus and all the filled inner shell electrons. Periodic trend of valence electrons All elements in a group have the same number of valence electrons equal to the first digit of their group number. It is very helpful to model a couple of electron-dot examples to get students started. On the other hand, the ionic radius is half the distance between two ions that barely touch each other in a compound. Periodicity in Properties of Elements The higher the effective nuclear charge Zeff, the more attracted the outer electrons are to the nucleus and the closer. 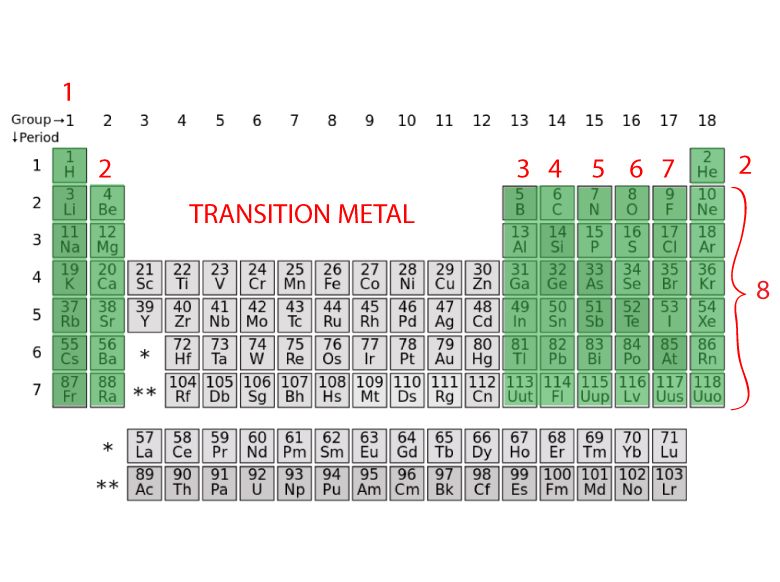
Atomic radius is the distance between an atom’s nucleus and its outermost or valence electrons. 
6. Properties of Elements Based on Periodic Trends 1. 6.10: The Explanatory Power of the Quantum-Mechanical Model The chemical properties of elements is determined primarily by the number and distribution of valence electrons. 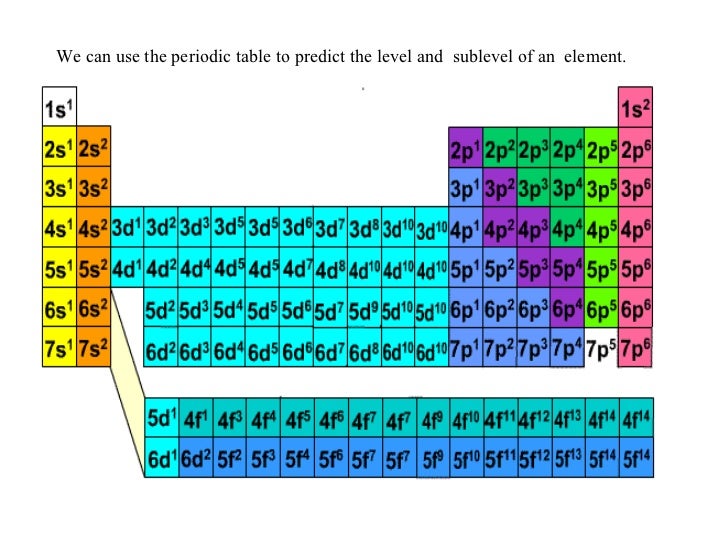
Adding the atomic numbers in the lower corner allows for the addition of mass numbers or atomic weights later if desired. Electron configurations can be predicted by the position of an atom on the periodic table. It maybe helpful to have students number the boxes one through twenty (atomic numbers) in the lower left corner first to avoid confusion. In this short activity (20-25 minutes) students will be provided a blank periodic table and will fill in the electron-dot model for the first twenty elements. This activity allows students to make a tool of models to help them visualize concepts of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
